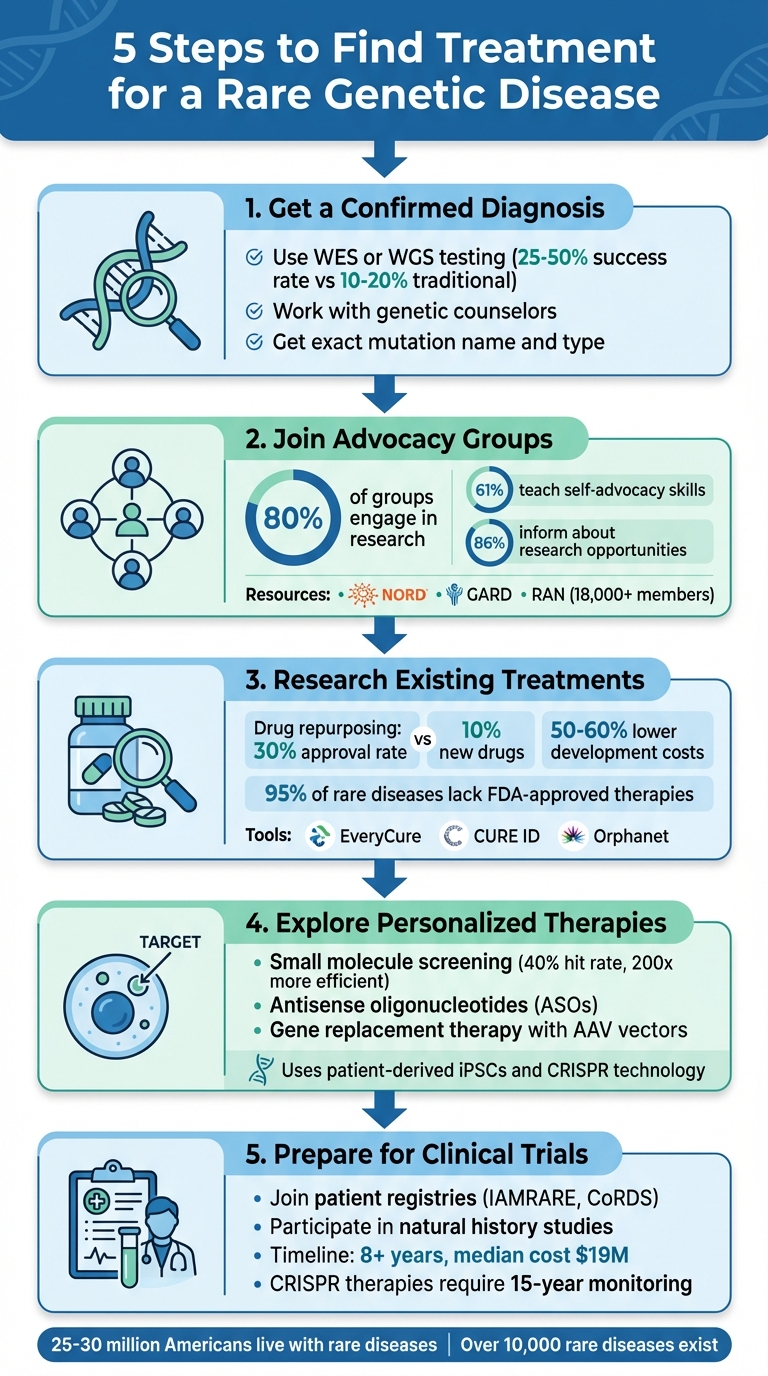
5 Steps to Find Treatment for a Rare Genetic Disease
A 5-step plan to pursue treatment for rare genetic diseases: confirm diagnosis, join advocacy groups, research repurposed drugs, explore personalized therapies, and prepare for trials.

Finding treatment for a rare genetic disease can be overwhelming, but there are actionable steps to help you navigate the process. Here's a quick guide:
- Get a Confirmed Diagnosis: Use advanced genetic testing like WES or WGS to identify the exact mutation causing the condition. Work with genetic counselors and specialists to interpret results and explore treatment options.
- Join Advocacy Groups: Connect with patient organizations for support, resources, and updates on research or clinical trials. Groups like NORD and GARD are great starting points.
- Research Existing Treatments: Look into drug repurposing, which applies FDA-approved medications to new conditions. Tools like EveryCure and Orphanet can help identify potential options.
- Explore Personalized Therapies: Programs like RareLabs use your genetic data and cells to develop tailored treatments, including gene therapy and small-molecule drugs.
- Prepare for Clinical Trials: Participate in registries and natural history studies to increase eligibility. Review trial criteria carefully and stay informed about preclinical progress.
These steps provide a clear path forward, from diagnosis to potential treatment. With advancements in genetic research and personalized medicine, there’s hope for addressing even the rarest conditions.
5 Steps to Find Treatment for Rare Genetic Diseases
Genetic editing to cure rare disease explained | 101 in 101
Step 1: Get a Confirmed Diagnosis with Genetic Testing
Genetic testing is a powerful medical tool used to identify changes in your DNA that may cause or increase the risk of certain diseases. For rare conditions, it can provide a definitive diagnosis, cutting down the often lengthy and frustrating diagnostic journey.
A confirmed genetic diagnosis does more than just name the condition - it opens doors to targeted treatments like gene therapies, helps you find relevant clinical trials, and informs your family about potential risks. Traditional genetic testing methods have a success rate of only 10–20%, but advanced techniques like whole exome sequencing (WES) and whole genome sequencing (WGS) can raise that rate to 25–50% or even higher.
Types of Genetic Testing Available
There are several types of genetic tests, each suited to different scenarios:
- Single gene testing: Focuses on one specific gene and is ideal when symptoms strongly suggest a particular condition, like sickle cell disease.
- Genetic panels: Examine multiple genes at once and are useful when several genes might be involved, such as in epilepsy cases.
- Whole exome sequencing (WES): Scans all protein-coding genes, which make up less than 2% of the genome but account for about 85% of known disease-causing mutations.
- Whole genome sequencing (WGS): Analyzes the entire genome, including non-coding regions, and is typically used when WES doesn’t yield answers. However, WES is more likely to be covered by insurance.
Thanks to next-generation sequencing technology, the cost and time required for these tests have dropped significantly. What used to take months or years now often takes just days or weeks.
Working with Genetic Counselors and Specialists
Genetic testing isn’t something you can do on your own - it requires a healthcare provider to order the test for a specific medical reason. Start by consulting your primary care provider, who can refer you to specialists like medical geneticists, neurologists, or hematologists. Genetic counselors are essential to this process. They help you choose the right test based on your symptoms and family history and guide you through understanding the results and their implications for you and your family.
Andrea Knob, a Genetic Research Counselor at Beth Israel Deaconess Medical Center, explains:
"The genetic counselor is there to provide information, options, and support."
To find qualified professionals, check out resources like the National Society of Genetic Counselors or the American College of Medical Genetics and Genomics. Before proceeding, confirm your insurance coverage by providing details like the test name, the performing lab, and the CPT codes to understand any potential costs. Some advocacy groups and pharmaceutical companies even offer free testing to reduce financial barriers. For those who remain undiagnosed after standard testing, the NIH’s Undiagnosed Diseases Network (UDN) connects patients with advanced technologies and specialists.
Using Test Results to Learn About Your Disease
Your genetic counselor or medical geneticist will explain the type of mutation involved - whether it’s autosomal dominant, autosomal recessive, X-linked, or de novo (a new mutation not inherited from parents).
Make sure to get the exact name and spelling of your diagnosis, including any subtypes or specific mutations. Understanding whether the mutation is inherited or de novo can help you assess risks for other family members. It’s also a good idea to keep a binder or digital folder with all your test results, appointment summaries, and insurance documents for easy access in the future.
Use the precise name of your mutation to search reliable resources like the Genetic and Rare Diseases Information Center (GARD) or the National Organization for Rare Disorders (NORD). You can also request a copy of your raw genetic data, which might be helpful for future research or second opinions.
A negative result doesn’t rule out all genetic disorders - it simply means no disease-causing mutation was found in the genes tested. Large-scale tests may also reveal secondary findings, such as risks for adult-onset conditions. As Gary Jackson, Senior Clinical Account Manager at Illumina, points out:
"Genetics are not the whole picture - just part of the picture."
After receiving a diagnosis, consider joining a patient registry like the NORD Registry Program or CoRDS. These networks can keep you informed about clinical trials and allow you to contribute to ongoing research. With a clearer understanding of your condition, you’re better equipped to connect with advocacy groups and others who share similar experiences.
Step 2: Join Patient Advocacy Groups
Once you've received a diagnosis, joining a patient advocacy group can make a world of difference. These groups provide not only emotional support but also reliable medical information and access to research opportunities. They can help transform feelings of isolation into informed action, empowering you to navigate your treatment journey with confidence.
What Advocacy Groups Offer
Patient advocacy groups go beyond offering a supportive community. Many play a critical role in advancing research and shaping policies for rare diseases. In fact, nearly 80% of these organizations actively engage in research by managing patient registries, conducting natural history studies, and educating healthcare professionals to improve treatment accessibility.
They also provide practical resources to directly support patients. For example, 61% of groups teach self-advocacy skills, helping individuals overcome insurance challenges and secure necessary therapies. Larger organizations often offer financial assistance programs that cover medication costs, insurance co-pays, and even travel expenses for clinical trial participation.
Rachel Gomel from PSC Partners Seeking a Cure sums up the impact of these groups:
"It always energizes me to get involved and do more. For lack of time, we often can't be present in all the wonderful projects NORD creates; however, knowing that they are there, and that we have a wide context to work from is always empowering."
The National Organization for Rare Disorders (NORD) is a key player in this space, operating the Rare Action Network (RAN). With over 18,000 members, RAN works on both local and national levels to influence federal policies affecting the 30 million Americans living with rare diseases. Advocacy groups also connect members with medical specialists, host educational webinars, and share updates on new research findings. In fact, 86% of these organizations actively inform their members about research opportunities.
Finding the Right Advocacy Group
To find the advocacy group that best fits your needs, start with specialized directories like the NORD "Find a Patient Organization" directory or the Genetic and Rare Diseases Information Center (GARD). These resources categorize groups by specific conditions, making it easier to locate the right support network. You can also use search engines to look up your specific gene or condition name along with terms like "advocacy", "support", or "foundation".
Social media platforms, especially Facebook, are another great way to connect. Many families and individuals create active support communities centered around shared experiences. Searching for your gene name alongside the word "support" can help you find these groups. If your condition is extremely rare and no formal organization exists, platforms like MyGene2.org allow you to connect with others who share your genetic variant.
When choosing an organization, prioritize those that are transparent about their funding sources and ensure their medical information is reviewed by qualified experts or a medical advisory board. Non-commercial groups that offer free, accurate information and disclose their funding sources are often the most trustworthy. If you’re unsure where to start, NORD’s helpline at 800-999-6673 is a valuable resource for immediate connections and guidance.
Additionally, consider joining patient registries like NORD's IAMRARE program or CoRDS. These registries help ensure you're included in future research studies and clinical trials. Your genetic counselor or specialist can also recommend reputable organizations tailored to your specific diagnosis.
Step 3: Research Existing Treatments and Drug Repurposing
After gathering insights from your advocacy network, the next step is to investigate existing medications that could be repurposed. Drug repurposing involves taking FDA-approved medications and applying them to new conditions. This method leverages prior research and safety data, making it a faster and more cost-effective alternative to developing new drugs from scratch.
How Approved Drugs Can Be Repurposed
Drug repurposing works by identifying medications that target biological pathways shared by different diseases. Since these drugs already have established safety profiles, they can reach patients more quickly and at a lower cost. For context, repurposed drugs achieve a 30% market approval rate, compared to just 10% for new drug applications, and they typically cost 50% to 60% less to develop.
Several databases and tools can help identify repurposing opportunities:
- EveryCure: Uses AI to analyze data from PubMed, clinicaltrials.gov, and medical records to find new applications for existing drugs.
- CURE ID: A collaboration between the FDA and NCATS, this platform allows clinicians to report real-world outcomes when using drugs off-label for rare diseases.
- Orphanet: Offers a comprehensive inventory of drugs, including those with orphan designation or marketing approval for rare diseases.
There are plenty of success stories highlighting the potential of drug repurposing. For example, Fenfluramine, originally used as an appetite suppressant, was repurposed for Dravet syndrome after clinical trials showed it could reduce seizure frequency by 63% to 72%. It was approved by the FDA and European Commission in 2020. Another example is propranolol, a cardiovascular drug that received FDA approval in 2014 for treating infantile hemangioma after doctors noticed its unexpected effectiveness during heart treatment.
When considering whether a repurposed drug might work for your condition, focus on whether your disease shares biological targets with the drug's original purpose. Genetic mutations play a key role too, as some drugs are effective only for specific mutations within the same gene. Patient-derived cell testing can help determine if a drug restores normal cellular function before committing to treatment. With 95% of rare diseases lacking FDA-approved therapies, repurposing offers a critical path forward.
Managing Symptoms with Nutrition and Supportive Care
While drug repurposing may provide targeted treatment, managing symptoms remains an essential part of care. Supportive measures can help ease daily challenges, maintain overall health, and reduce complications related to your condition. These approaches also prepare you for potential clinical trials by improving your baseline health.
Specialists familiar with your specific condition can guide you in managing symptoms. For example, genetic metabolic disorders often require tailored nutrition plans to prevent toxic metabolite buildup or address enzyme deficiencies. Therapies like physical, occupational, and speech therapy can also help maintain function and independence.
Your advocacy group can be an invaluable resource, connecting you with others who have practical strategies for symptom management. Many groups also maintain registries of specialists with experience in supportive care for your condition. Keeping a detailed record of what works and what doesn’t can help you communicate effectively with your medical team and contribute valuable information to researchers studying your condition.
Step 4: Consider RareLabs' Personalized Drug Discovery Program
When repurposed drugs fall short, personalized drug discovery offers a tailored solution. RareLabs takes a patient-specific approach, using your own cells to identify therapies designed for your exact genetic mutation. This method is particularly valuable given the staggering reality: there are over 10,000 rare diseases, yet most patients have no access to targeted treatments. Personalized drug discovery steps in where standard methods fail, creating new possibilities.
How RareLabs' Program Works
The process begins with collecting biological samples such as skin, muscle, or blood to create a unique cell line. RareLabs then uses whole genome sequencing (WGS) to pinpoint the exact breakpoints of your mutation, which can sometimes reveal unexpected diagnostic details. Skin biopsy cells are converted into induced pluripotent stem cells (iPSCs), providing a renewable resource for testing.
To refine their approach, RareLabs employs CRISPR/Cas9 technology to correct the mutation in some of the patient-derived cells. For example, in a study focused on Retinitis Pigmentosa, CRISPR successfully corrected 13% of RPGR gene copies in iPSCs. These corrected cells act as controls during therapeutic screening, helping researchers identify treatments that restore normal cellular function.
Therapeutic screening involves in-vitro assays to test whether potential treatments can restore protein levels or function. RareLabs also uses advanced AI platforms like BioNAV™ to analyze thousands of compounds, achieving a remarkable 40% hit rate - making it 200 times more efficient than traditional random screening methods. For certain conditions, in-vivo models, including CRISPR-engineered organisms, are used to evaluate the effects of treatments on motor function, seizures, or cognition.
Treatment Options at RareLabs
RareLabs offers multiple therapeutic strategies based on your specific mutation and disease mechanism. Here’s a breakdown:
- Small Molecule Screening: This approach identifies FDA-approved drugs that could be used off-label for your condition, leveraging patient-specific cell testing.
- Antisense Oligonucleotides (ASOs): These small synthetic genetic molecules target specific RNA sequences and are particularly effective for neurodegenerative disorders.
- Gene Replacement Therapy: Using next-generation Adeno-Associated Virus (AAV) vectors, this method delivers functional copies of the affected gene directly into your cells.
RareLabs also includes a Personalized Modality Assessment Amenability Plan, which determines the best therapeutic approach for your unique mutation. In some cases, combination therapies are recommended to address multiple disease mechanisms at once. To ensure the integrity of the process, quality checks like karyotyping are conducted to confirm the genetic stability of your cell lines.
Staying Updated on Your Program Progress
RareLabs keeps you informed every step of the way. They provide updates that break down complex scientific findings into clear, understandable information you can share with your medical team. Milestone reports track progress from the establishment of your cell line to therapeutic screening and validation testing. You’re encouraged to ask questions, ensuring you fully understand how each discovery relates to your condition. This transparency helps you make informed decisions and prepares you for the next stages, such as preclinical assessments and potential clinical trials.
Step 5: Move Forward with Preclinical Models and Clinical Trials
Once potential therapies are identified, the next step involves rigorous testing to ensure they are safe and effective before moving to human trials. Preclinical models serve as a critical bridge between laboratory discoveries and clinical trials, helping researchers evaluate safety, dosage, and efficacy. This phase is crucial - 90% of drugs that make it to clinical trials fail, often due to unforeseen toxicity or inefficacy that earlier tests didn't reveal.
Building Disease Models from Patient Cells
Patient-derived cells offer a way to recreate a "disease in a dish." By reprogramming skin, blood, or urine cells into induced pluripotent stem cells (iPSCs), scientists can generate disease-relevant cell types like neurons or heart muscle cells. Unlike animal models, these human-cell models are better at replicating the symptoms seen in patients.
These models allow researchers to study how therapies impact DNA, RNA, and protein levels specific to your genetic makeup. In addition, advanced techniques like 3D organoids and "organs-on-a-chip" simulate complex tissue environments, such as the fluid dynamics in lungs or intestines. The FDA has even acknowledged the power of these approaches. For example, they expanded approval for the cystic fibrosis drug ivacaftor to cover 23 additional mutations based solely on in vitro data from cell models. This decision significantly sped up access for patients with rare mutations.
"Animal models often provide poor insights into disease mechanisms of rare diseases." - Frontiers in Cell and Developmental Biology
If you're providing tissue samples for preclinical testing, timing is critical. For instance, muscle biopsies should come from a muscle that shows symptoms but hasn't yet atrophied too much. Severely damaged tissue often fails to establish viable cell lines. Once the cell lines are created, whole genome sequencing ensures they reflect your clinical diagnosis and accurately pinpoint mutation breakpoints.
With these validated models in hand, researchers can move toward clinical trials.
Getting Ready for Clinical Trials
After preclinical testing confirms safety and efficacy, the focus shifts to clinical trials. This process can take eight years or more from start to finish, with a median cost of $19 million. To get involved, consider joining patient registries like NORD's IAMRARE or CoRDS. These databases collect health and genetic information, helping researchers find and recruit suitable participants.
Participation in natural history studies is another key step. These studies track how a disease progresses without treatment, providing critical baseline data for designing effective clinical trials. A compelling example comes from Taylor's Tale, a non-profit that funded $350,000–$400,000 for preclinical research on CLN1 and CLN2 (Batten disease) at the University of North Carolina at Chapel Hill. This work, led by Dr. Steven Gray, transitioned to a biopharma company by 2016, received IND acceptance in 2019, and progressed to Phase 1/2 clinical trials.
Before enrolling in a trial, carefully review the eligibility criteria. Factors like age, gender, specific genetic mutations, disease severity, and medical history often determine who qualifies. You can search for active studies and eligibility details on platforms like ClinicalTrials.gov or the Gene Therapy Trial Browser. It's also wise to schedule pre-IND meetings with the FDA to clarify trial design and endpoint requirements. For CRISPR-based therapies, be aware that the FDA mandates long-term monitoring - patients are followed for 15 years post-treatment to ensure safety.
Developing human-grade therapies for ultra-rare diseases often comes with a hefty price tag, exceeding $2 million. This is where patient advocacy groups and non-profit biotech organizations play a vital role. Groups like Global Genes and the EveryLife Foundation provide resources to help you navigate the complexities of drug development and connect with researchers seeking trial participants.
Conclusion
Finding a treatment for a rare genetic disease is far from a solo endeavor. The five steps in this guide outline a clear path, starting with confirmed genetic testing to secure the ICD code necessary for insurance approvals and clinical trial eligibility. Partnering with patient advocacy groups and registries can also help increase your visibility to researchers. While awaiting long-term solutions, exploring existing treatments and drug repurposing options can help manage symptoms in the interim.
This structured approach - moving from diagnosis to customized drug discovery - provides a way to take charge of your treatment journey. With 95% of rare diseases still lacking FDA-approved therapies, personalized medicine offers hope. RareLabs' program, for instance, uses patient-derived cells to create precision therapies tailored to specific genetic mutations, even for those affecting only a handful of individuals worldwide.
"The journey to treatment-ready status begins with that first, small step you take today." - William Sarraille, Founder, Sarraille & Associates
As genetic research continues to advance rapidly, it's crucial to stay in touch with your specialist and stay informed about emerging treatment options. Preclinical models and clinical trials often serve as the critical final steps, offering access to experimental therapies that may otherwise be unavailable. These pathways are vital for addressing conditions that impact 25 to 30 million Americans.
The road from diagnosis to effective treatment is becoming more navigable. With precise diagnoses, strong community connections, and personalized discovery programs, the potential for effective treatments is closer than ever.
FAQs
How do I know if I need WES or WGS?
When deciding between Whole Exome Sequencing (WES) and Whole Genome Sequencing (WGS), your specific diagnostic goals play a key role.
WES zeroes in on coding regions of the genome, which are frequently associated with genetic disorders. This makes it a targeted approach, often quicker and less expensive. On the other hand, WGS examines the entire genome, including both coding and non-coding regions, offering a broader and more detailed genetic picture. This can be especially helpful in cases where the cause of a condition is unclear or when a more exhaustive analysis is needed.
For the best course of action, it's always a good idea to consult a genetic specialist who can help determine which option aligns with your needs.
What should I do if my genetic test is negative?
A negative genetic test result indicates that no alterations were detected in the specific gene, chromosome, or protein that was analyzed. However, it’s important to note that this result doesn’t entirely eliminate the possibility of a genetic disorder. To gain a clearer understanding of what this means for your health, it’s a good idea to discuss the findings with a genetic counselor or healthcare provider.
If symptoms continue or new ones develop over time, it may be worth exploring additional testing or seeking specialized medical guidance. Another option to consider is participating in research studies or clinical trials, which could provide further insights into your condition and potential next steps.
How can I improve my chances of getting into a clinical trial?
To improve your chances of being part of a clinical trial for a rare disease, begin by checking out clinical trial databases and connecting with patient advocacy groups. These resources can help you identify opportunities and provide vital support. Talk to your healthcare provider or geneticist for personalized guidance and potential referrals. Stay active in your search by keeping an eye on emerging treatments like gene or cell therapies. Additionally, engage with specialized research centers and organizations that share updates and offer assistance tailored to your condition.



