What to Ask Before Starting Rare Disease Treatment
Questions to ask before starting rare disease treatment—confirm genetic diagnosis, compare therapies, weigh risks and costs, and plan long-term follow-up.

Starting treatment for a rare disease is a complex decision. Here's what you need to know upfront:
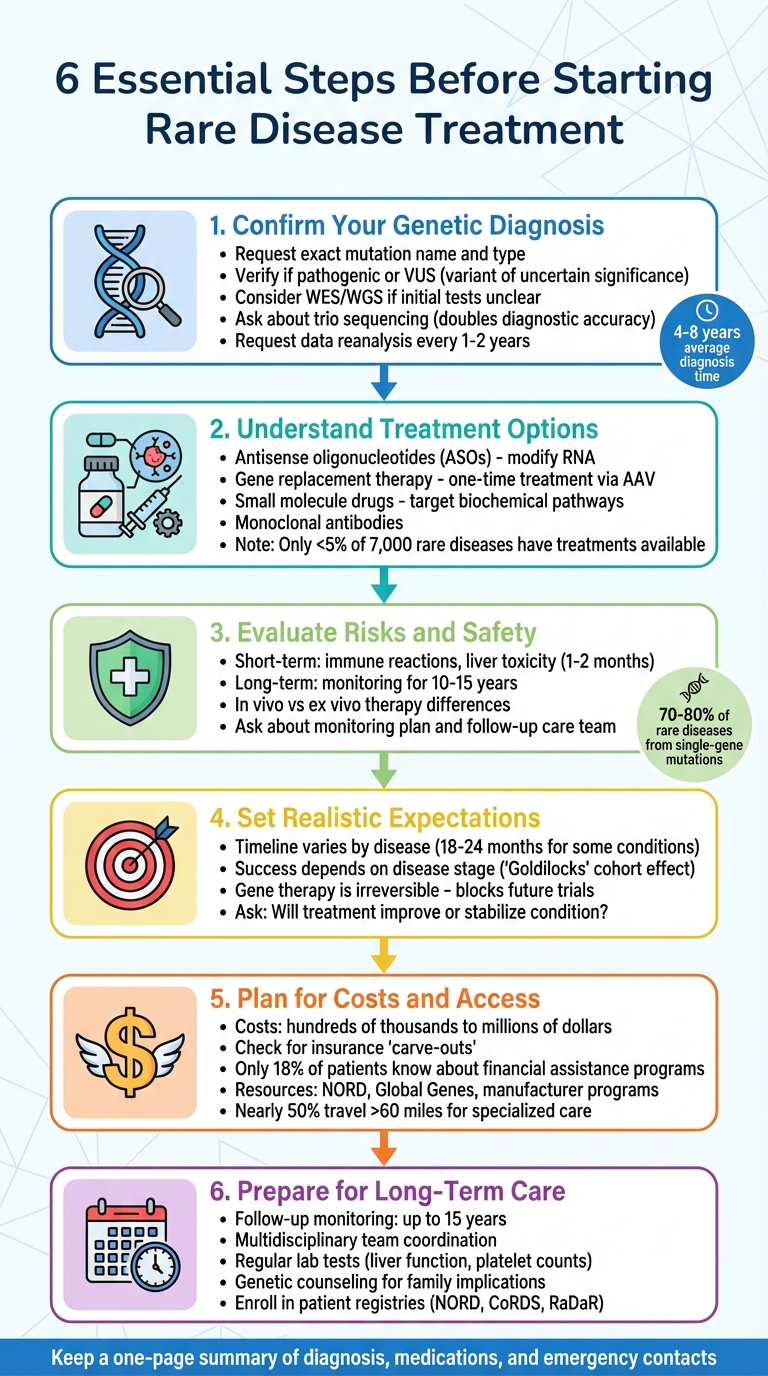
- Confirm Your Genetic Diagnosis: Ensure your diagnosis is accurate and complete. Ask about your specific mutation, its classification (pathogenic or uncertain), and additional testing options like whole-exome sequencing (WES) or whole-genome sequencing (WGS).
- Understand Treatment Options: Treatments vary by mutation type. Options include gene therapy, antisense oligonucleotides (ASOs), and small molecule drugs. Each has unique benefits, risks, and timelines.
- Evaluate Risks and Safety: Short-term risks might include immune reactions or liver toxicity. Long-term risks, like potential complications over years, require monitoring. Ask about follow-up care and safety protocols.
- Set Realistic Expectations: Treatments may stabilize or improve your condition, but outcomes depend on disease stage and therapy type. Clarify success metrics and timelines with your doctor.
- Plan for Costs and Access: Rare disease treatments can be expensive. Check insurance coverage, financial assistance programs, and patient support resources to manage costs.
- Prepare for Long-Term Care: Treatment often involves ongoing monitoring, genetic counseling, and coordination with specialists. Ensure you're ready for follow-ups that could last years.
6 Essential Steps Before Starting Rare Disease Treatment
Verify Your Genetic Diagnosis and Profile
Before starting any treatment, it's essential to ensure your genetic diagnosis is both accurate and thorough. Genetic disorders stem from mutations that disrupt protein production, impacting cellular processes and causing symptoms. The specific mutation identified can determine the severity of your condition and how it may progress over time.
Most rare diseases have a genetic basis, but getting an accurate diagnosis can take anywhere from 4 to 8 years on average. This delay often happens because initial genetic tests might not provide definitive answers. A negative result on a specific gene panel doesn’t necessarily mean there aren’t mutations in genes that weren’t included in that panel.
"If the test finds a rare variant in one of those genes, suddenly that may seem significant but might not be the answer."
- Dr. Brent Fogel, MD, PhD, FAAN, Director of the UCLA Clinical Neurogenomics Research Center
Understanding whether your mutation is classified as a known "pathogenic" variant, which is confirmed to cause disease, or a "variant of uncertain significance" (VUS) is a critical step before deciding on treatment. If your results reveal a VUS, ask if patient-derived models can be used to verify whether the mutation is actually responsible for your condition.
Questions About Genetic Testing Results
Start by requesting the exact name and type of genetic mutation identified in your lab report. Ask your healthcare provider whether the mutation is known to cause disease or falls into the VUS category. Another helpful question is, "What symptoms are typical for my mutation at my age?" This can help determine if your symptoms align with those typically associated with the identified mutation.
If the initial test results are unclear, inquire about whole-exome sequencing (WES) or whole-genome sequencing (WGS), and consider requesting trio sequencing. Trio sequencing, which tests you alongside both biological parents, has been shown to double diagnostic accuracy while significantly reducing the number of possible variants.
"The diagnostic probability of the trio approach was twice as high as that of single tests."
- Dr. Hakan Gürkan, Department of Medical Genetics, Trakya University
Additionally, ask if your genetic data will be reanalyzed every 1–2 years. As global research advances, variants initially classified as uncertain may later be reclassified with new evidence. Consulting a certified genetic counselor can also help you interpret complex results and explore any implications for your family.
If uncertainties persist, patient-derived models may provide further clarity about your diagnosis.
Patient-Derived Models and Testing
For cases involving a VUS or inconclusive test results, ask about the possibility of using patient-derived models to study your condition further. These models often involve reprogramming your cells into induced pluripotent stem cells (iPSCs) to investigate how your mutation impacts cellular function.
Functional studies using both in vitro and in vivo models can help determine whether a novel variant is genuinely causing your disease, while also revealing the underlying molecular mechanisms. For instance, RareLabs develops patient-specific iPSC models and CRISPR-corrected controls to confirm genetic diagnoses and evaluate potential therapies tailored to your mutation. This method can provide a clearer understanding of whether a treatment is likely to work for your unique genetic profile.
If WES or WGS results don’t yield answers, consider asking about additional analyses, such as transcriptomic, proteomic, or metabolomic studies, to identify hidden abnormalities.
Review Treatment Options and Methods
Once your genetic diagnosis is confirmed, it's time to explore treatment strategies tailored to your specific mutation. The right approach depends heavily on the type of genetic alteration involved.
What Treatment Methods Are Available
There are several therapeutic options for rare genetic conditions, each targeting different mutation types. One approach involves antisense oligonucleotides (ASOs), which are chemically engineered to modify RNA. These can either reduce harmful proteins or restore missing ones. ASOs come in different forms, such as:
- Gapmers: These recruit enzymes to break down toxic mRNA.
- Splice-switching ASOs: These adjust RNA splicing to help produce normal proteins.
Another option is gene replacement therapy, which introduces a functional copy of a gene into cells. This is often done using viral vectors like adeno-associated viruses (AAV) and is typically a one-time treatment, unlike ASOs, which require regular doses every 1 to 4 months.
Small molecule drugs and monoclonal antibodies offer additional options by targeting specific biochemical pathways. For example, Alhemo works by blocking a protein that inhibits clotting in Hemophilia A.
Currently, disease-modifying treatments are available for fewer than 5% of the 7,000 known rare diseases. However, since 70–80% of rare conditions stem from single-gene mutations, many are candidates for emerging gene-targeted therapies. It’s essential to discuss with your healthcare provider which treatments have been studied for your specific mutation. Whether your condition involves a gain-of-function or loss-of-function mutation will guide the choice of treatment.
Matching Treatment to Your Condition
Once you understand the available methods, the next step is identifying which one best suits your genetic profile. The effectiveness of a treatment depends on the specific characteristics of your genetic variant. For example:
- Exon skipping with splice-switching ASOs can address in-frame variants.
- Personalized ASO therapies may target deep intronic variants, depending on eligibility criteria.
Your healthcare team can help determine if your mutation has been assessed using standardized guidelines like the N1C VARIANT framework, which categorizes genetic variants as "eligible", "likely eligible", or "unlikely eligible" for ASO treatment.
A Personalized Modality Assessment can also evaluate your genetic profile against available therapies. This assessment considers key factors, including your mutation type, inheritance pattern, and whether your gene can tolerate protein knockdown.
For conditions involving toxic gain-of-function variants, where reducing protein levels is the goal, it’s crucial to confirm that your gene can handle such downregulation. If the gene is haploinsufficient (requiring at least 50% normal activity), excessive knockdown could inadvertently cause a loss-of-function disorder. To ensure the treatment is effective and safe for your mutation, ask about functional studies using patient-derived models. These studies can provide critical insights into how well a therapy might work for you.
Learn About Risks, Side Effects, and Safety
When exploring treatment options, it's crucial to understand the risks and safety considerations associated with each one. Every treatment comes with potential risks, and being informed early on can help you prepare for what to expect. For newer treatments, like gene therapy, some risks may only come to light over time. As Dr. Amy Waldman points out, "Gene therapy carries both short-term and long-term risks that individuals and families should understand before participating in clinical trials".
Short-Term and Long-Term Risks
In the initial three months following treatment, an overactive immune response may lead to symptoms such as vomiting and general discomfort. Liver toxicity is another serious concern, often appearing within one to two months. This may require treatment with steroids to manage the condition. To minimize inflammation and protect the liver, healthcare providers typically use immunosuppressants.
The long-term risks are harder to predict. Since gene therapies are designed to have lasting effects from a single dose, patients are monitored for 10 to 15 years to detect delayed complications, including the potential for cell overgrowth or cancer. Another area of uncertainty is how long the treatment's benefits will persist. Dr. Eric Crombez, Chief Medical Officer at Ultragenyx, explains:
"Because gene therapies are intended to provide long-term treatment with a single dose, study participants are monitored for years".
Ask your healthcare provider about your specific monitoring plan. Will you be part of a patient registry to track long-term safety? Who will oversee your follow-up care after the initial treatment phase? Monitoring typically involves a team that includes your primary care doctor, a specialist for your condition, and a gene therapy expert.
Safety Differences Between Treatment Types
The method used to deliver the treatment plays a significant role in its safety profile. For in vivo therapies, the treatment is administered directly through an IV infusion. The primary risks include immune reactions to the viral vector (such as AAV) and liver toxicity. These are generally managed with steroids. However, one notable limitation of in vivo therapy is that after the first dose of AAV, the immune system "remembers" it, making repeat doses of the same therapy impossible.
On the other hand, ex vivo therapies involve removing your cells, modifying them in a laboratory, and then reintroducing them into your body. This approach often requires chemotherapy beforehand to clear out existing cells, which can bring its own set of intensive side effects and risks. Additionally, patients may need to stay in a hospital isolation room while their immune system recovers and the modified cells settle in. Be sure to confirm whether chemotherapy or other preparatory steps are necessary and understand the associated risks.
| Treatment Type | How It's Given | Main Safety Concerns | How Risks Are Managed |
|---|---|---|---|
| In Vivo | Direct injection or IV infusion | Immune response to viral vector; liver toxicity | Steroids to reduce inflammation |
| Ex Vivo | Cells removed, modified, and returned to the patient | May require chemotherapy; hospital isolation due to reduced immune function | Monitoring for cell engraftment and immune system recovery |
Set Expectations for Outcomes and Timelines
Gene therapies are often described as one-time treatments, but their full effects can take years to unfold. Before starting, it’s crucial to understand what success might look like for your condition and how long it could take to see results. The timeline varies significantly depending on the disease, its progression, and the treatment method.
Success Rates and Outcome Variability
Setting realistic expectations is just as important as considering safety. Timing plays a critical role in the success of gene therapy. You need to be healthy enough to undergo the treatment but also affected enough to show measurable improvement - a concept researchers call the "Goldilocks" cohort effect. If your disease is too advanced, the therapy may not reverse existing damage, though it could help prevent further decline. A key question to ask your doctor is: Am I at a stage where treatment could improve my condition, or is the goal to stabilize it?
Success is measured differently depending on the disease. For example, in spinal muscular atrophy, doctors assess whether a child can sit unsupported for 30 seconds, typically over 18 to 24 months. For hemophilia A or B, they track annualized bleeding rates and factor activity levels over a period of 52 weeks to two years. In some cases, biomarkers are used to evaluate progress. For instance, the FDA’s 2023 approval of Tofersen for SOD1-associated ALS highlighted how biomarkers like neurofilament light chain levels can indicate treatment efficacy, even if immediate clinical improvements aren’t evident.
An important consideration is that gene therapies are irreversible. If a treatment is ineffective, it may prevent you from participating in future clinical trials or accessing new therapies. As noted in Advances in Therapy:
"It is essential that for any gene therapy on the market, there is reasonable certainty that the treatment will be efficacious before treatment is given because once given, the therapy cannot be taken away and currently removes the possibility of the patient being treated with another gene therapy or getting into other clinical trials".
How Progress Will Be Tracked and Reported
Once you’ve established potential outcomes, tracking progress through clear metrics and timelines becomes vital. Progress is often measured using Clinical Outcome Assessments (COAs) - tests designed for specific conditions and symptoms. These might include mobility tests, enzyme levels, protein production, or bleeding rates. For instance, in Duchenne muscular dystrophy, a meaningful change could be a 30-meter improvement on the six-minute walk test. For conditions affecting cognitive function, wearable biometric sensors are now used to monitor gait, coordination, and sleep patterns without requiring active participation from the patient.
Talk with your healthcare team about which biomarkers or functional milestones will define success for you. Ask for a clear timeline: When should I expect to see the first signs of improvement? You’ll likely have follow-ups for up to 15 years, comparing your progress to natural history data through patient registries.
These assessments, combined with your personalized treatment plan, help ensure that your progress aligns with safety and treatment goals.
Understand Costs, Access, and Support
Treatment Costs and Insurance Coverage
Gene therapies come with hefty price tags, often ranging from hundreds of thousands to several million dollars. And that’s just the starting point. The listed price usually doesn’t include expenses like administration, hospital stays, or follow-up care. On top of that, diagnostic testing - sometimes spanning years - can rack up significant costs even before treatment begins.
When dealing with insurance, it’s essential to dig into the fine print. Ask your provider if your prescribed medication is subject to "carve-outs", which are exclusions that might leave certain orphan drugs uncovered, even under comprehensive private insurance plans. It’s also worth requesting a case manager from your insurer to help estimate your total out-of-pocket expenses. Rare disease treatments tend to be three to five times more expensive than those for more common conditions.
Here’s something not everyone knows: only about 18% of patients are aware that pharmaceutical companies offer financial assistance programs. If you’re looking for help with medical bills, travel, or other non-covered costs, organizations like the National Organization for Rare Disorders (NORD) or disease-specific groups may have grants available. And if your insurance denies coverage, you might still be eligible for assistance from manufacturer-sponsored patient foundations - often without needing to formally appeal the denial. Some programs, such as the Evrysdi Starter Program, even provide an initial free 30-day supply (with the possibility of one refill) while you wait for insurance approval.
Understanding these costs is just the first step - next comes exploring the resources available to help you navigate this challenging journey.
Available Support Resources
Managing the financial burden is tough, but ensuring you have access to the right support services is equally important. Beyond costs, you’ll want to connect with organizations and resources that can guide you through treatment logistics.
Groups like Global Genes' RARE Concierge and NORD's Information & Resource Services are excellent places to start. They can help you find disease-specific specialists and Centers of Excellence, and they offer these services at no cost. Since nearly half of rare disease patients travel over 60 miles to access specialized care, it’s also worth checking out travel assistance programs offered by non-profits.
For those enrolled in personalized drug discovery programs, such as those provided by RareLabs, the level of support is unmatched. You’ll get direct access to a team of scientists and clinicians who guide you throughout your treatment. This includes regular updates in plain language, milestone tracking, and the opportunity to ask questions directly to the experts working on your case. With this kind of hands-on support, you’re not left to navigate complex decisions alone. Instead, you’ll have a team coordinating care across specialists, labs, and treatment centers, ensuring every aspect of your journey is managed with care and precision.
Plan for Treatment Start and Long-Term Care
Eligibility Requirements and Preparation
Before starting treatment, meeting specific eligibility criteria is crucial. These steps ensure that the therapy aligns with your unique genetic profile and is administered safely. For example, a genetically confirmed diagnosis, such as bi-allelic SMN1 mutations for spinal muscular atrophy, is often required. Other factors include appropriate anti-AAV9 antibody titers (at or below 1:50 for viral-vector therapies), stable baseline evaluations (like liver function, creatinine levels, and blood counts), and the absence of active infections. Proper hydration and nutrition are also essential.
Certain therapies may require pausing medications, like hydroxyurea, for 2–3 months. As Dr. Rao, a neuromuscular specialist, highlights:
"The earlier the treatment, the better the outcome".
Administrative preparation is just as important. Complete all necessary insurance forms, patient support documents, and informed consent paperwork. A social worker or patient advocate can help manage these logistics. For ex vivo therapies, expect hospital stays of 1–3 days for cell collection, with possible repeat sessions spaced about four weeks apart. Some treatments also involve conditioning phases, such as starting steroids the day before infusion or undergoing chemotherapy to prepare the bone marrow.
It’s a good idea to ask your healthcare team for a visual roadmap of the treatment process, including follow-up visit timelines. Early coordination with specialists - such as pediatric gastroenterologists, hematologists, or cardiologists - can also streamline the process.
Once the eligibility and preparation steps are complete, the focus shifts to long-term follow-up and counseling.
Follow-Up Care and Genetic Counseling
After treatment begins, long-term follow-up becomes a priority. For advanced therapies like gene therapy, monitoring can last up to 15 years to assess effectiveness and identify any delayed side effects. Regular lab tests will be necessary to evaluate liver function, platelet counts, and cardiac health. For example, patients receiving Zolgensma must have liver function and platelet levels monitored for at least three months until stable results are achieved.
Care coordination with a multidisciplinary team is crucial. This team might include medical geneticists, neurologists, and condition-specific specialists, such as hematologists or cardiologists. As Geoffrey Beek, a certified genetic counselor, explains:
"Geneticists, doctors who specialize in genetic disorders, are often very good at managing care for patients with complex rare diseases".
Staying organized is key. Keep a one-page summary of your diagnosis, medications, and emergency contacts handy. A dedicated binder for tracking appointments and test results across providers can also be helpful. Additionally, consider enrolling in disease-specific registries like NORD, CoRDS, or RaDaR. These registries not only keep you informed about evolving care standards but also contribute to valuable research. As GenePossibilities notes:
"In addition to follow-up care, you may be added to a patient registry. This registry helps gather important information that can help people who will receive gene therapy in the future".
Genetic counseling is another vital component. Schedule sessions with a genetic counselor to discuss recurrence risks and determine whether family testing is needed. Tools like the National Society of Genetic Counselors’ online directory can help you find local or telehealth professionals. Geoffrey Beek underscores the importance of this step:
"A genetic counselor can help you understand the genetic aspects of your child's diagnosis, what it means for your family, and whether testing is recommended for siblings or other relatives".
Finally, as Dr. Tesi Rocha reminds patients:
"Even if the patient looks great, they still need to adhere to those follow-up visits that we stipulated at the beginning of the treatment".
Conclusion
Before starting treatment, it’s essential to ask the right questions: confirm your specific genetic mutation, explore available treatment options, weigh potential risks, and set realistic expectations for outcomes.
Preparation is just as important as the treatment itself. Keep a running list of questions handy and create a one-page summary of your diagnosis, current medications, and emergency contacts for easy reference by specialists. Consider connecting with patient advocacy groups and registries like NORD or CoRDS to stay updated on clinical trials and emerging treatments.
Your level of preparedness can influence both short-term results and long-term care. As Geoffrey Beek, a certified genetic counselor, aptly puts it:
"Understanding your child's diagnosis is a journey, not a race".
By asking personalized questions and advocating for yourself or your loved one, you can ensure the treatment aligns with your genetic profile. While hundreds of gene therapies are in clinical trials, only a small number have received full FDA approval. Seeking second opinions from specialists who focus on your condition can make a significant difference in your care.
Progress stems from having access to trustworthy information, working with a coordinated multidisciplinary team, and building connections with others who share similar experiences with rare diseases. Taking an active role in your care equips you with the confidence to navigate treatment options effectively. With the right resources and questions, you can approach your treatment journey with clarity and determination.
FAQs
Should I get a second opinion on my genetic results?
Yes, getting a second opinion is often a smart move, especially when dealing with complex or life-altering diagnoses. It can help confirm your initial results, give you peace of mind, and ensure that you've explored all possible treatment options. By consulting other specialists or genetic counselors, you can gain a clearer understanding of your results, evaluate potential risks, and tailor treatments to fit your specific genetic makeup.
How do I know which therapy matches my mutation type?
To find the most suitable therapy for your specific mutation, it's essential to consult with a healthcare provider or genetic specialist. They can carefully review your genetic test results and assess whether your mutation qualifies for targeted treatments, such as exon skipping or splice-switching therapies. Be sure to inquire about the most up-to-date clinical guidelines and eligibility requirements to confirm the treatment is appropriate for your condition and mutation type.
What could make me ineligible for treatment right now?
If you don’t meet certain criteria - like age, genetic profile, current health condition, or how you’ve responded to past treatments - you might not qualify for the treatment. Typically, eligibility is determined through tests or assessments conducted during the screening process to confirm that the treatment is both safe and suitable for you.



