8 Questions About Patient-Derived Cell Models Answered
How patient-derived iPSCs, organoids, and PDX models improve drug discovery, personalized therapies, and their limits.

Patient-derived cell models are changing how we study rare genetic diseases, offering a closer look at patient-specific conditions. These models are created using cells, like blood or skin, from patients and reprogrammed into induced pluripotent stem cells (iPSCs). Unlike older methods, these models retain the complete genetic profile of the individual, including mutations and modifiers that affect disease progression and treatment response.
Key takeaways:
- What they are: Models made from patient cells, such as iPSCs, organoids (3D cell structures), and xenografts (tumors implanted in mice).
- Why they matter: They mimic patient biology, helping researchers understand diseases better and test treatments more accurately.
- How they’re made: Patient cells are reprogrammed into stem cells using non-integrating methods, ensuring safety and precision.
- Applications: Drug discovery, testing gene therapies, and personalized treatment validation.
- Challenges: Issues with reproducibility, scalability, and cost remain hurdles.
These models are already advancing treatments, including FDA-approved gene therapies and personalized drug testing, but they still face limitations like high costs and production complexity.
What Are Patient-Derived Cell Models?
Definition and Purpose
Patient-derived cell models are specialized tools crafted from the actual cells or tissues of a patient. Unlike standard lab-grown cell lines, these models originate from samples such as blood, skin, or tumor biopsies taken directly from individuals with a particular disease. Researchers then use these samples to create functional models for in-depth study.
What sets these models apart is their ability to retain the patient’s complete genetic makeup. This means they include not only the primary mutation causing the disease but also other genetic factors that influence how the disease manifests and progresses. For rare genetic conditions, where traditional models often fall short, patient-derived models offer a more accurate way to study the disease in a relevant biological context.
There are several types of patient-derived models, each serving a unique purpose:
- Patient-derived iPSCs: Induced pluripotent stem cells reprogrammed from patient tissues.
- Organoids: Three-dimensional structures grown from patient tissue that replicate real organ architecture.
- Patient-derived xenografts (PDX): Tumor tissues from patients implanted into immunodeficient mice, where they grow while maintaining their original characteristics.
This genetic fidelity makes these models invaluable for studying patient-specific disease conditions.
How They Replicate Disease Conditions
Because they preserve the patient’s complete genetic profile, these models offer an accurate snapshot of the disease as it exists in the body. Patient-derived models act as "faithful replicas" of the original tissue, capturing the intricate genetic, protein, and cellular features that define the disease. Unlike engineered cell lines, which typically introduce a single mutation into a healthy genetic background, these models reflect the full complexity of multifactorial diseases.
Three-dimensional models, like organoids, go even further by replicating not just the genetics but also the physical environment of diseased tissue. This includes factors like nutrient availability, cellular interactions, and structural organization. As noted by Greiner Bio-One:
Tumoroids can recapitulate the complex genetic and molecular compositions of solid cancers, making them extremely valuable in the study of disease progression.
This detailed replication helps explain why two individuals with the same rare disease may experience vastly different symptoms or respond differently to treatments. Their unique genetic backgrounds create distinct disease profiles, which patient-derived models can effectively capture and analyze.
Engineering Organoid Models for Cancer Research
How Are Patient-Derived iPSCs Created?
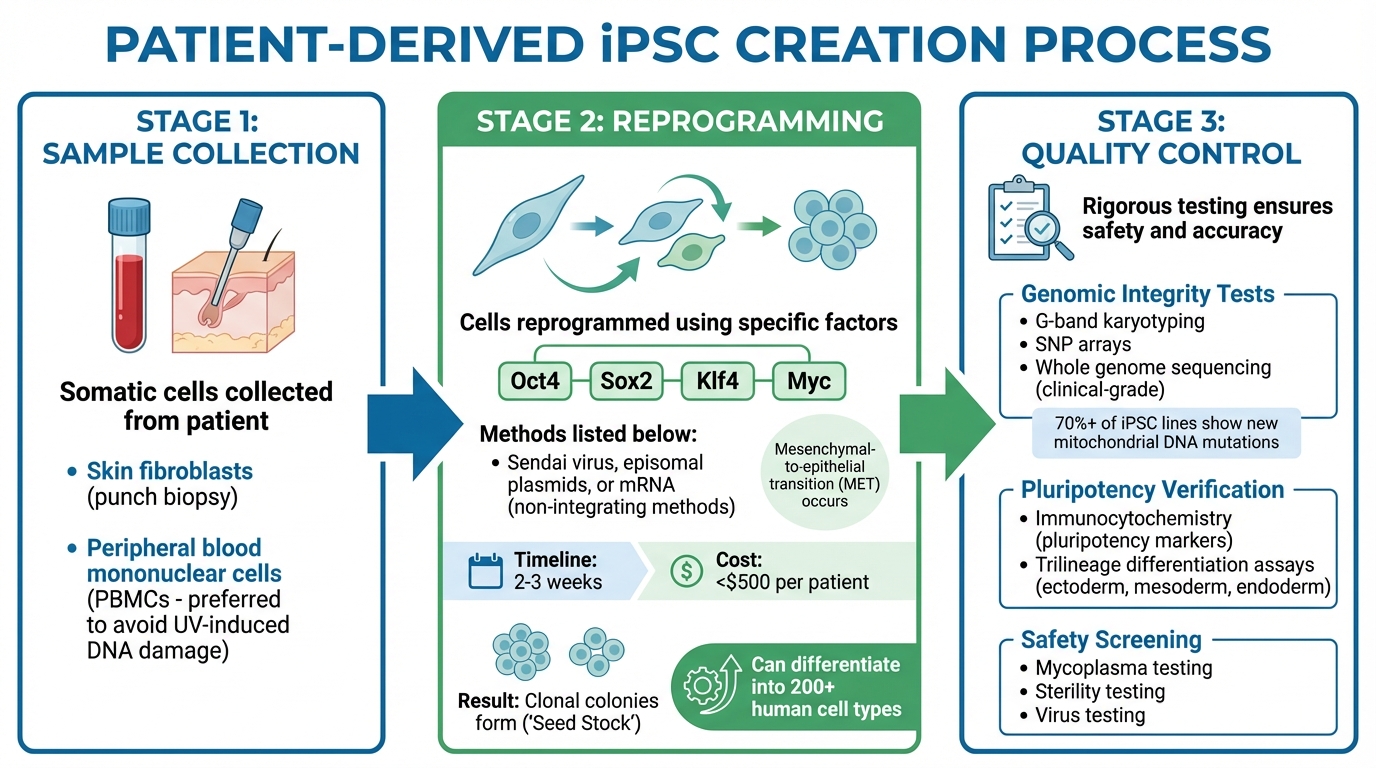
How Patient-Derived iPSCs Are Created: From Sample Collection to Quality Control
Sample Collection and Reprogramming
The creation of patient-derived iPSCs starts with collecting somatic cells from the patient. Common sources include skin fibroblasts, obtained through a punch biopsy, and peripheral blood mononuclear cells (PBMCs). PBMCs are often preferred because they avoid the risk of ultraviolet-induced DNA damage.
To reprogram these cells, specific factors - Oct4, Sox2, Klf4, and Myc - are introduced using non-integrating methods like Sendai virus, episomal plasmids, or mRNA. These approaches are ideal for clinical applications because they don't leave behind foreign genetic material. During this process, cells undergo molecular changes, such as a mesenchymal-to-epithelial transition (MET). This transition causes fibroblasts to adopt the compact, rounded shape typical of stem cells. Once reprogrammed, the cells form clonal colonies, referred to as "Seed Stock", which can be expanded and differentiated into more than 200 human cell types.
After reprogramming, stringent quality control ensures the iPSCs accurately reflect the patient's genetic profile.
Quality Control in iPSC Creation
Quality control is critical to ensure patient-derived iPSCs are safe, functional, and reliable. Genomic integrity is evaluated using methods like G-band karyotyping, SNP arrays, and, for clinical-grade lines, whole genome sequencing. Interestingly, studies have found new iPSC-specific mitochondrial DNA mutations in over 70% of iPSC lines.
In addition to genomic checks, scientists confirm the cells have reached true pluripotency. Techniques like immunocytochemistry are used to detect key pluripotency markers, while trilineage differentiation assays verify the cells can form all three germ layers - ectoderm, mesoderm, and endoderm. Safety screenings also test for contaminants, including mycoplasma, sterility, and viruses.
As The Jackson Laboratory highlights:
To maintain genomic integrity and reduce unintended editing effects, institutions like JAX employ advanced gene-editing protocols coupled with rigorous quality control measures.
These thorough quality control steps ensure that iPSCs serve as reliable tools for research. By transforming patient samples into accurate models of an individual's genetic makeup, iPSCs play a key role in advancing personalized drug testing and therapy development.
How Are Patient-Derived Cell Models Used in Drug Discovery?
Patient-derived cell models play a key role in advancing drug discovery for rare genetic conditions. These models replicate human disease mechanisms, offering a real-world cellular environment to study diseases and evaluate potential treatments. By using cells with the exact genetic mutations found in patients, researchers can better understand disease pathways and identify effective therapies. With over 300 million people globally affected by rare diseases - and more than 90% of these conditions lacking treatments - these models are opening up new possibilities.
Screening Potential Treatments
Once patient-derived iPSCs (induced pluripotent stem cells) are differentiated, they become powerful platforms for high-throughput drug screening. Specialized assays can then pinpoint compounds that restore normal cellular functions.
One striking example is a primary liver cancer biobank created between 2019 and 2024 by Hui Yang and colleagues. This biobank included 399 tumor organoids derived from 144 patients. Researchers used these organoids to test drug responses and discovered that c-Jun signaling was responsible for resistance to lenvatinib, a widely used liver cancer drug. By combining lenvatinib with a c-Jun inhibitor, they demonstrated a strong synergistic effect that overcame the resistance. This personalized approach to drug screening would have been nearly impossible with traditional animal models.
These advancements in drug screening are now laying the groundwork for more precise therapeutic validation.
Testing Gene and RNA-Based Therapies
Patient-derived cell models are essential for testing cutting-edge treatments such as antisense oligonucleotides (ASOs), RNA-based therapies, and gene replacement strategies. These therapies require rigorous validation in human cells before they can proceed to clinical trials.
A groundbreaking example is the FDA's December 2025 approval of Waskyra (etuvetidigene autotemcel), the first gene therapy for Wiskott-Aldrich syndrome. Developed by Fondazione Telethon ETS, this therapy involves harvesting a patient’s hematopoietic stem cells, using a lentiviral vector to insert a functional WAS gene, and reinfusing the corrected cells. This ex vivo gene therapy showed remarkable results in clinical trials with 27 patients, reducing severe infections from 2.0 to 0.2 per patient-year. Patient-derived cell models were instrumental in refining this therapy, ensuring effective genetic correction and proper cell functionality.
Using CRISPR-Corrected Controls
One of the challenges in drug discovery is determining whether a treatment addresses the underlying mutation or is influenced by patient-specific genetic factors. CRISPR-Cas9 technology offers a solution through CRISPR-corrected controls. Researchers use this tool to create "revertant lines", where the disease-causing mutation in patient-derived iPSCs is corrected back to its healthy sequence.
These corrected cells, known as isogenic controls, have the same genetic background as the original patient-derived cells, with the only difference being the corrected mutation. This allows researchers to directly compare the effects of treatments in a controlled genetic environment. As The Jackson Laboratory notes:
Using isogenic sets allows researchers to isolate and compare the effects of different variants within the same genetic background... providing an unprecedented level of control.
These examples highlight how patient-derived models not only accelerate drug discovery but also refine personalized treatment strategies for rare diseases.
What Are Patient-Derived Organoids and How Are They Used?
What Are Organoids?
Patient-derived organoids (PDOs) are three-dimensional models grown from a patient’s own stem or tumor cells. Often called "mini-organs", these lab-grown structures replicate the original tissue’s architecture, function, and genetic characteristics. Unlike traditional 2D cell cultures, organoids include multiple cell types, making them more representative of actual human tissues.
The process of creating organoids starts with collecting a tissue sample through methods like needle biopsy, surgical resection, or biological fluid extraction. The sample is broken down into single cells or small clusters, which are then embedded in a 3D gel scaffold - commonly Matrigel. These cells are cultured with a growth medium enriched with factors like EGF, Wnt3a, and Noggin, tailored to the tissue type. Within about two weeks, the cells multiply and differentiate, forming small organ-like structures.
This ability to mimic real tissue makes organoids a powerful tool for drug testing, as explored in the next section.
How Organoids Are Used in Drug Testing
Thanks to their tissue-like complexity, patient-derived organoids are transforming how researchers evaluate drug efficacy and safety, especially for rare genetic conditions where traditional models often fail. It’s worth noting that nearly 92% of cancer drugs fail during clinical trials, largely due to the limitations of conventional testing methods.
Organoids allow for personalized drug testing, offering a more accurate prediction of how a specific patient might respond to a treatment. Srinivas Maddi of Acubiosys Private Limited highlights this advantage:
The major benefit of 3D organoid models is their ability to predict drug candidate efficacy and toxicity, significantly impacting the reduction of attrition rates in drug research and development.
For example, a clinical trial (NCT04254705) conducted in February 2020 by Universitaire Ziekenhuizen Leuven tested the effectiveness of ivacaftor/tezacaftor in 30 cystic fibrosis patients carrying the rare R334W mutation. By using organoids derived from rectal biopsies, researchers could predict which patients were most likely to benefit from the treatment.
PDOs are also used in safety testing. In a 2026 study, researchers applied 3D bioprinting to create kidney organoids with precise size and cell counts. These models were used to test aminoglycosides for toxic effects, revealing selective toxicity in specific cell types, such as kidney and lung cells. Another advantage of PDOs is their ability to be cryopreserved and stored in "living biobanks", making them a long-term resource for ongoing drug testing, even years after the original biopsy was taken.
What Are the Benefits of Patient-Derived Cell Models?
Better Prediction of Drug Response
Patient-derived cell models offer a more precise way to predict how individual patients will respond to treatments compared to older methods. Traditional immortalized cell lines, which have been cultured for decades, often lose the unique genetic and molecular traits of a patient's disease. In contrast, short-term cultured patient-derived cells (PDCs) retain these characteristics, preserving the genomic and molecular diversity of the original tumor.
"Models derived from short-term–cultured patient-derived cells (PDCs) retain the genomic characteristics of solid tumor biopsy better than immortalized cell lines."
For example, between December 2016 and February 2020, researchers at the National Cancer Center in Korea developed 102 patient-derived cell models from individuals with advanced lung cancer. By testing these models against 48 anti-cancer compounds, they discovered that XAV939 was effective for patients resistant to osimertinib. Additionally, they found that the PI3K-AKT inhibitor alpelisib was able to inhibit growth in SCLC PDC models, a result that wasn't observed when using standard immortalized cell lines.
One of the standout advantages of PDC models is their ability to uncover resistance mechanisms. They allow researchers to identify specific molecular subtypes that are likely to resist certain treatments, enabling the selection of alternative therapies before beginning treatment. The study also highlighted the efficiency of these models, achieving a 92.2% success rate in establishing PDC models from pleural effusions across 102 lung cancer samples. This approach is not only effective but also minimally invasive, making it an ideal method for studying advanced stages of disease.
This predictive capability paves the way for highly personalized therapeutic strategies.
Enabling Customized Therapies
Patient-derived models go beyond predicting drug responses - they play a crucial role in crafting personalized treatment plans. By capturing the complete disease profile, including primary mutations and genetic modifiers, these models provide the foundation for precise drug screening and tailored therapies.
They also account for individual genetic variations that influence how a disease progresses and presents symptoms. This is especially critical for rare genetic conditions, where traditional models often fail to replicate the unique clinical features of each patient. By testing treatments directly on patient cells, researchers can identify promising drug candidates using high-throughput screening methods.
These advantages highlight how patient-derived models are transforming the way rare genetic diseases and other complex conditions are treated, offering a more individualized and effective approach to care.
What Are the Limitations of Patient-Derived Cell Models?
While patient-derived cell models offer valuable insights, they come with several challenges that can limit their effectiveness in research and drug development.
Reproducibility Challenges
One of the biggest obstacles with patient-derived iPSC models is maintaining consistency across experiments. iPSC-derived organoids often face difficulties due to lengthy differentiation protocols and uneven maturation, which can make reproducibility a serious issue. Unlike adult stem cells, which are already specialized for a particular organ, iPSCs need precise external cues - like Wnt, FGF, and TGFβ pathways - to differentiate into the desired cell types.
The problem becomes even more complex with "unguided" differentiation methods. In these cases, hiPSC aggregates undergo spontaneous morphogenesis, which can lead to a wide variety of cell types within a single sample. This unpredictability adds significant variability to experiments. As Elisa Heinzelmann and her team at Doppl SA observed:
Challenges such as prolonged differentiation protocols and variability in maturation levels remain significant hurdles.
To tackle these issues, researchers are turning to more controlled techniques. Guided differentiation processes, which use specific growth factors and small molecules, can help achieve more consistent outcomes. Tools like multi-well spinning bioreactors are also being used to produce uniform structures, such as cortical layers in forebrain organoids. Additionally, leveraging validated hiPSC lines from international biobanks can minimize the variability that often arises when generating cell lines in individual labs. However, these variability concerns also make scaling up these models particularly difficult.
Scalability and Cost Issues
Creating and maintaining patient-derived iPSC lines is both expensive and time-consuming, which limits their use in large-scale applications. The costs associated with generating, validating, and sustaining complex 3D cultures are significant. Each cell line requires customized protocols, making it hard to standardize processes for high-throughput screening. Moreover, the multi-step differentiation process adds to the time and expense compared to simpler models based on adult stem cells.
Genetic variability between donors adds another layer of complexity. These differences can mask the effects of specific mutations, forcing researchers to use larger sample sizes to achieve statistically meaningful results - further driving up costs. To address these financial and logistical challenges, institutions are adopting advanced quality control techniques. For example, long-range PCR is used to confirm genetic edits, SNP arrays help detect copy number variations, and karyotyping ensures chromosomal stability. These measures help maintain quality but do little to reduce the overall expense of these models.
Conclusion
Patient-derived cell models are reshaping the landscape of personalized medicine, especially for rare genetic diseases where conventional treatments are limited or unavailable. These models, such as induced pluripotent stem cells (iPSCs) and organoids, mimic patient biology, making them invaluable tools for preclinical therapy testing. This approach, often referred to as "functional testing", has shown impressive predictive accuracy, with studies reporting 81% sensitivity and 74% specificity in predicting treatment outcomes.
Advancements in technology have significantly improved the efficiency and affordability of these models. For instance, new techniques now enable the production of patient-derived iPSCs in just 2 to 3 weeks at a cost of less than $500 per patient - a dramatic improvement from the traditional process, which could take up to a year and cost between $5,000 and $10,000. Dr. Scott Younger from Children's Mercy highlights the importance of these developments:
The widespread ability to generate patient-derived cellular systems will have a substantial effect on the understanding of disease mechanisms as well as potential therapeutic avenues for the treatment of many rare diseases.
While challenges around reproducibility and scalability remain, these models are already having a tangible impact. In January 2025, the Children's Mercy Kansas City Genomic Medicine Center used patient-derived organoids to validate personalized antisense oligonucleotide treatments for three patients with Duchenne muscular dystrophy. Within eight weeks, this approach successfully restored dystrophin protein function. These advancements underscore the role of patient-derived cell models as indispensable tools in the development of tailored therapies.
FAQs
How is my consent and privacy handled when my cells are used to make a model?
Your consent and privacy are protected through a clear and transparent informed consent process. This ensures you’re fully aware of how your cells will be utilized. Additionally, robust privacy policies are implemented to safeguard your personal information during the development and use of patient-derived models.
How long does it take to create a patient-derived iPSC line or organoid for testing?
Creating a patient-derived iPSC line or organoid is a time-intensive process, typically requiring several weeks to a few months. The timeline can vary based on the specific protocols used and the overall complexity of the procedures involved.
How well do patient-derived models predict real treatment response in people?
Patient-derived models offer a reliable way to predict how treatments might work. Unlike conventional cell lines, these models closely mimic the complexity of tumors and human biology. This makes them especially useful for tailoring therapies to individual patients and testing new drugs. By better reflecting real-world conditions, they enhance the chances of success in precision medicine.



