Gene Therapy vs Antisense Oligonucleotides: Key Differences
Compare gene therapy and antisense oligonucleotides: DNA vs RNA targets, durable vs reversible effects, delivery methods, and best-use cases for rare genetic diseases.

Gene therapy and antisense oligonucleotide (ASO) therapy are two cutting-edge approaches to treating rare genetic disorders. Here’s what you need to know:
- Gene Therapy: Works at the DNA level to replace, edit, or silence faulty genes. It often uses viral vectors (like AAV) for delivery and aims for long-lasting or permanent effects, typically requiring a single dose.
- ASO Therapy: Targets RNA to influence protein production. ASOs are synthetic molecules that degrade harmful RNA, modify splicing, or block protein production. Effects are temporary, requiring repeated treatments.
Key Differences
- Target: Gene therapy focuses on DNA; ASOs act on RNA.
- Duration: Gene therapy effects are long-term; ASOs are short-term and reversible.
- Delivery: Gene therapy uses viral vectors; ASOs rely on chemically modified oligos.
- Applications: Gene therapy is ideal for loss-of-function mutations, while ASOs excel in addressing toxic gain-of-function mutations or splicing defects.
Quick Comparison
| Feature | Gene Therapy | Antisense Oligonucleotides (ASOs) |
|---|---|---|
| Primary Target | DNA | RNA |
| Mechanism | Gene replacement/editing/silencing | RNA degradation/splicing/blocking |
| Duration | Long-lasting (one-time dose) | Temporary (repeated dosing) |
| Delivery | Viral vectors (e.g., AAV) | Synthetic oligos or conjugates |
| Administration | IV or localized injection | Intrathecal or systemic |
| Best For | Loss-of-function mutations | Toxic gain-of-function mutations |
Both therapies have unique strengths and challenges, with the choice depending on the genetic mutation, delivery needs, and treatment goals.
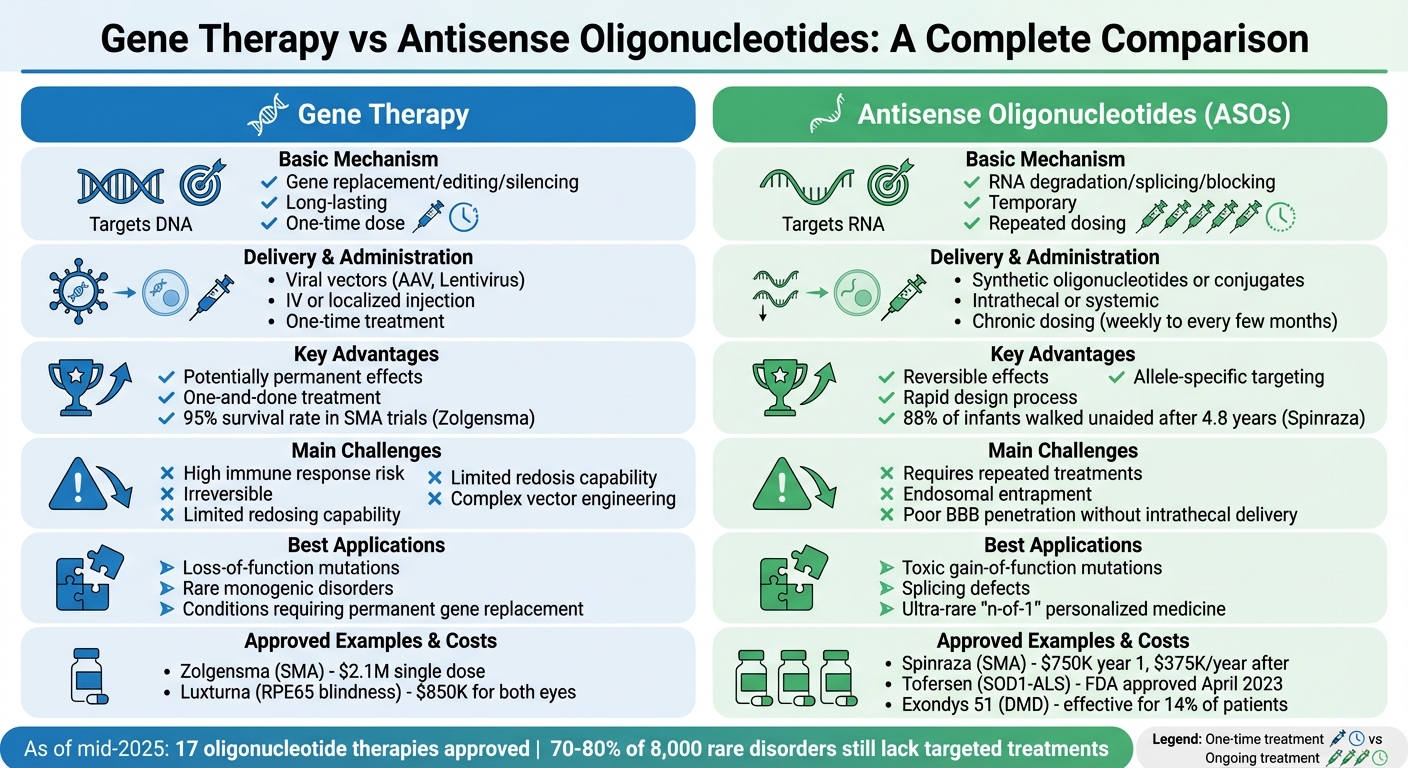
Gene Therapy vs Antisense Oligonucleotides Comparison Chart
How Gene Therapy and Antisense Oligonucleotides Work
Gene Therapy
Gene therapy operates at the DNA level, aiming to tackle genetic issues by altering the genome or introducing new genetic material for long-term effects. It employs three main approaches: augmentation, editing, and silencing.
Gene augmentation involves delivering a functional copy of a gene (cDNA) into cells to make up for a missing or defective version. This is often done using viral vectors like adeno-associated virus (AAV) or lentivirus, which transport the genetic material to the nucleus. A good example is Onasemnogene abeparvovec (Zolgensma), a treatment for spinal muscular atrophy. It uses an AAV9 vector to deliver a working copy of the SMN1 gene through a single intravenous infusion, showing its effectiveness in managing rare conditions.
Gene editing uses molecular tools like CRISPR/Cas9, zinc-finger nucleases, or TALENs to make precise changes, deletions, or corrections to DNA sequences. On the other hand, gene silencing uses viral vectors to deliver instructions, such as shRNA or microRNA, that suppress the expression of harmful mutant genes. Since gene therapy targets DNA, its effects are designed to be long-lasting, often requiring just a single treatment. These methods are particularly promising for treating rare genetic disorders.
While gene therapy focuses on DNA for lasting corrections, ASO therapy takes a more flexible approach by targeting RNA.
Antisense Oligonucleotides (ASOs)
Unlike gene therapy's DNA-based approach, ASO therapy works at the RNA level, adjusting gene expression without altering DNA. ASOs are short, single-stranded synthetic molecules (15–30 base pairs) that bind to specific RNA sequences through Watson-Crick base pairing. To improve their stability and binding strength, ASOs are chemically modified with features like phosphorothioate backbones, MOE, or LNA.
ASOs work through three main mechanisms: RNase H-mediated degradation, splice modulation, and translation inhibition.
- RNase H-mediated degradation uses "gapmer" ASOs, which have a DNA core flanked by modified RNA regions. When these ASOs bind to target mRNA, they form a DNA:RNA hybrid that activates RNase H, an enzyme that degrades the transcript. For instance, Tofersen (Qalsody), approved by the FDA in April 2023 for SOD1-related ALS, reduces levels of the toxic mutant protein through this mechanism.
- Splice modulation relies on steric block ASOs that bind to pre-mRNA, masking splice-regulatory elements. This can either skip problematic exons to restore the reading frame or include missing exons to produce functional proteins. Nusinersen (Spinraza), approved in December 2016, targets the SMN2 gene to promote exon 7 inclusion, increasing the production of functional protein in spinal muscular atrophy patients. Similarly, Eteplirsen (Exondys 51), approved in September 2016, skips exon 51 of the DMD gene, helping restore the reading frame in certain Duchenne muscular dystrophy cases.
- Translation inhibition prevents protein synthesis by physically blocking the ribosome from binding to or moving along the mRNA. Since ASOs cannot cross the blood-brain barrier, treating central nervous system disorders requires intrathecal administration - direct injection into the spinal fluid. This RNA-focused method allows for highly targeted therapies tailored to specific genetic profiles.
Side-by-Side Mechanism Comparison
Here’s a quick look at how these two approaches differ:
| Feature | Gene Therapy | Antisense Oligonucleotides (ASOs) |
|---|---|---|
| Primary Target | DNA (Genome) or long-term cDNA expression | RNA (mRNA or pre-mRNA) |
| Mechanism | Gene replacement, insertion, or editing | RNase H degradation, splice modulation, or steric block |
| Duration of Effect | Potentially permanent or long-lasting (one-time treatment) | Transient/Reversible (requires repeated dosing) |
| Delivery Vehicle | Viral vectors (e.g., AAV, Lentivirus) | Synthetic naked oligos or chemical conjugates |
| Administration | Often intravenous or localized injection | Intrathecal (for CNS) or systemic (subcutaneous/IV) |
| Reversibility | Irreversible post-integration | Effects wear off as ASO degrades |
Delivery Methods and Administration
Gene Therapy Delivery
Gene therapy primarily relies on viral vectors to deliver genetic material into target cells. Among the most commonly used are adeno-associated virus (AAV) and lentivirus, chosen for their ability to effectively enter cells and transfer therapeutic genes. The delivery approach varies based on the disease and affected tissues. Some therapies involve a single intravenous infusion, while others require direct injections into specific areas, such as the eye via subretinal delivery.
That said, viral delivery isn't without its challenges. Pre-existing immunity can interfere with treatment - many individuals already have neutralizing antibodies against AAV due to prior natural exposure, which can block the therapy's effectiveness. Additionally, once a patient undergoes gene therapy, their immune system typically prevents redosing with the same vector. AAV vectors also have a limited capacity to carry large genes, making them unsuitable for certain applications. Achieving precise tissue targeting is another obstacle, requiring advanced vector engineering for better specificity.
ASO Delivery
Unlike viral vectors, antisense oligonucleotides (ASOs) employ a non-viral delivery method to reach their targets. These are administered as chemically modified naked oligonucleotides or conjugated to targeting ligands.
Chemical modifications play a key role in ensuring ASOs survive within the body. For instance, phosphorothioate backbones and sugar modifications like 2'-MOE protect ASOs from enzymatic degradation and extend their half-life. Some 2'-MOE ASOs can persist in cerebrospinal fluid for more than six months. Targeted delivery has also improved, thanks to conjugation strategies. For example, attaching GalNAc (N-acetylgalactosamine) to ASOs boosts their liver-targeting efficiency by 10 times compared to their unconjugated counterparts, enabling them to bind specifically to hepatocyte receptors.
However, ASOs face their own hurdles, particularly with cellular uptake and endosomal escape. A significant portion of ASOs that enter cells becomes trapped in endosomes - cellular compartments that degrade or export their contents - preventing the ASOs from reaching their RNA targets in the cytoplasm or nucleus. This "endosomal trap" is a major barrier to therapeutic success and remains a focus of ongoing research.
Delivery Method Comparison
| Feature | Gene Therapy | ASOs |
|---|---|---|
| Delivery Vehicle | Viral vectors (AAV, Lentivirus) | Naked/modified oligonucleotides or lipid nanoparticles |
| Administration Routes | Intravenous, subretinal | Subcutaneous, intravenous, intrathecal, intravitreal |
| Treatment Frequency | One-time dose | Chronic dosing (weekly to every few months) |
| Immune Response Risk | High (neutralizing antibodies to vector) | Lower (primarily innate immune activation) |
| BBB Penetration | Poor; requires direct CNS injection | Poor; requires intrathecal delivery |
| Targeting Precision | Dependent on viral tropism | Enhanced by chemical conjugates (e.g., GalNAc) |
| Redosing Capability | Limited by immune response | Possible with repeated administration |
Applications in Rare Genetic Diseases
Gene Therapy Use Cases
Gene therapy is transforming the treatment landscape for rare diseases by replacing defective genes. Let’s look at some real-world examples that highlight its potential. Zolgensma (onasemnogene abeparvovec) has become a game-changer for spinal muscular atrophy (SMA). This one-time intravenous treatment achieved an impressive 95% survival rate in clinical trials, far surpassing other therapies. However, its price tag - $2.1 million for a single dose - reflects its groundbreaking nature, as it eliminates the need for ongoing treatments.
Another standout is Luxturna (voretigene neparvovec), which addresses RPE65-related blindness. Administered via subretinal injection, this therapy costs $425,000 per eye (or $850,000 for both). Patients have maintained improved vision for at least five years following treatment. Luxturna works by delivering a functional copy of the RPE65 gene directly to retinal cells, enabling them to produce the protein essential for vision.
"The hope and promise of gene therapy is that it could just be a one-time therapy that would alleviate the need for bi-weekly treatments." - Sarah Neuhaus, DO, Senior Medical Director, Prevail Therapeutics
While gene therapy offers the prospect of a single, lasting treatment, RNA-targeted therapies provide an alternative approach for ongoing disease management.
ASO Use Cases
Antisense oligonucleotides (ASOs) take a different approach, focusing on RNA to manage diseases through repeated dosing. These therapies are designed to precisely target RNA, making them ideal for addressing specific genetic conditions. For example, Spinraza (nusinersen) is used to treat SMA by targeting RNA splicing to boost SMN protein production. In the NURTURE study, 88% of infants treated with nusinersen were able to walk unaided after 4.8 years of therapy. Spinraza requires intrathecal injections every four months, with costs of $750,000 in the first year and $375,000 annually thereafter.
For Duchenne muscular dystrophy (DMD), Exondys 51 (eteplirsen) employs exon-skipping to restore the reading frame of the dystrophin gene. This therapy is effective for the 14% of DMD patients with mutations amenable to exon 51 skipping, demonstrating the precision of ASOs in targeting specific genetic variants. Additionally, in April 2023, the FDA approved Tofersen for SOD1-associated ALS. This ASO targets the mRNA responsible for producing a toxic mutant protein, effectively reducing its levels.
ASOs have also opened doors for "n-of-1" personalized medicine, where treatments are tailored for individuals with ultra-rare mutations. A striking example is Milasen, developed in 2018 for a 6-year-old girl with Batten disease. This custom therapy reduced her seizures from 15–30 per day to 0–20, with shorter episodes. Milasen underscores how ASOs can be rapidly designed for rare genetic conditions affecting just one or a few patients worldwide.
ASO versus AAV gene therapy strategies in Huntington’s disease
Strengths and Weaknesses of Each Approach
Building on the earlier discussion of mechanisms and delivery methods, let’s dive into the strengths and challenges of each therapeutic approach.
Gene Therapy: Pros and Cons
Gene therapy is often hailed for its long-lasting, sometimes one-time treatment potential. By replacing or supplementing defective genes, it can deliver sustained benefits. A great example is Zolgensma, which, in the START trial, showed effects lasting through a five-year extension. This "one-and-done" approach is especially impactful for rare monogenic disorders where a missing protein needs permanent replacement.
However, it's not without risks. Severe immune reactions to viral vectors are a concern, and the effects are irreversible if adverse outcomes occur. Additionally, the intricate process of vector engineering can slow down development compared to ASO therapies.
Antisense Oligonucleotides (ASOs): Pros and Cons
ASOs provide a highly targeted and reversible way to address specific RNA transcripts. This precision allows them to differentiate between mutant and normal alleles, making them particularly effective for toxic gain-of-function mutations while preserving healthy protein production. The reversibility is a safety advantage - if treatment stops, the effects gradually diminish.
"Perhaps the most significant advantage to ASO therapeutics over other small molecule approaches is that acquisition of the target sequence provides immediate knowledge of putative complementary oligonucleotide therapeutics."
– Daniel R. Scoles, PhD, University of Utah
Another strength of ASOs is their rapid design process, enabling personalized treatments, such as Milasen for Batten disease. On the downside, ASOs require ongoing treatments - Spinraza, for instance, needs intrathecal injections every four months. Delivery to areas like the central nervous system also remains a key challenge.
Pros and Cons Comparison
The table below highlights the key differences between these two approaches:
| Feature | Gene Therapy | Antisense Oligonucleotides (ASOs) |
|---|---|---|
| Durability | Long-term; often a single dose | Short-term; requires repeated dosing |
| Reversibility | Generally irreversible | Reversible; effects fade if treatment stops |
| Immune Response | High risk (viral vector reactions) | Lower risk; generally manageable |
| Design Speed | Complex vector engineering | Rapid; based on RNA sequence |
| Best For | Rare monogenic loss-of-function disorders | Toxic gain-of-function mutations, splicing defects |
| Precision | Broad gene replacement | Can target specific alleles or SNPs |
Clinical Progress and Future Directions
Gene therapy and antisense oligonucleotides (ASOs) are making strides, each with a distinct approach to treating genetic disorders. Gene therapy continues to grow its therapeutic scope, while ASOs are increasingly tailored for personalized treatments, offering solutions that align with individual genetic profiles.
Take "N-of-1" therapies as an example. These are treatments designed for a single patient, such as a custom ASO therapy developed for a KIF1A-associated disorder. In August 2024, researchers, including Dr. Wendy K. Chung from Boston Children's Hospital, reported a breakthrough: a patient with a KIF1A-associated neurological condition received a custom ASO targeting the p.Pro305Leu variant. After nine months of treatment, the patient experienced fewer behavioral arrest episodes and falls, while maintaining stable cognitive abilities.
Infrastructure programs like the "1 Million 1 Month" (1M1M) initiative and the N-of-1 Collaborative (N1C) are further streamlining the process from genetic discovery to personalized treatment. For instance, in April 2023, the FDA approved Tofersen for SOD1-associated ALS. This decision, based on its ability to reduce neurofilament light chain levels, showed regulatory flexibility by relying on surrogate biomarkers, even though some clinical endpoints were not fully met. This kind of adaptability is speeding up the approval process for treatments targeting fast-progressing diseases.
ASO therapies are also being explored in younger, more vulnerable populations. For example, preterm infants with severe developmental and epileptic encephalopathies are now receiving ASO treatments as early as seven weeks of age. In one notable case, an ASO therapy for SCN2A-related epilepsy reduced seizure frequency by more than 60%. As of mid-2025, the FDA had approved 17 oligonucleotide therapies, yet around 70–80% of the 8,000 known rare disorders - mostly caused by single-gene mutations - remain untapped targets for these approaches.
The future of gene therapy and ASO treatments looks promising, especially with advancements in delivery methods. Exosome-based carriers and lipid-albumin nanoparticles are expanding the reach of these therapies beyond the liver and central nervous system to include muscles and other tissues. While over 90% of rare diseases still lack targeted treatments, these innovations are opening doors to new possibilities. The challenge now lies not just in advancing the science but also in ensuring that patients can access these groundbreaking therapies as they move from research to real-world application.
Conclusion
Selecting the right therapy hinges on understanding the genetic cause and the required treatment timeframe. Gene therapy focuses on altering DNA, offering long-term or even permanent solutions. This makes it a strong option for loss-of-function conditions where a faulty or missing gene needs replacement. On the other hand, ASOs (antisense oligonucleotides) act on RNA, providing temporary and reversible effects. This flexibility can be particularly helpful for addressing toxic gain-of-function mutations or when safety concerns demand the ability to stop treatment quickly.
It's essential to identify whether the condition stems from a toxic gain-of-function or loss-of-function mechanism. For instance, ASOs are effective in reducing toxic mRNA in SOD1-associated ALS, while gene therapy is better suited for conditions caused by haploinsufficiency.
Delivery methods and the accessibility of target tissues also play a critical role. ASOs work well for localized treatments, such as intrathecal injections for the central nervous system or intravitreal injections for the eye. Gene therapy, often delivered via viral vectors, can target multiple tissues systemically. However, its use is typically limited to a single administration due to potential immune responses. These delivery considerations help refine the decision based on the affected tissues and logistical factors.
Clinicians must also consider the severity and progression of the disease. Fast-progressing conditions may justify higher risks, as demonstrated by the approval of Tofersen in April 2023. Additionally, ultra-rare diseases with unique mutations are increasingly benefiting from "n-of-1" customized ASO therapies, which can often be developed more quickly than traditional gene therapy approaches.
FAQs
Which patients should choose gene therapy vs ASOs?
Gene therapy is often best for patients dealing with severe, single-gene disorders. These conditions can benefit from a one-time, durable solution that corrects or replaces faulty genes. On the other hand, antisense oligonucleotides (ASOs) are more suitable for individuals with specific genetic mutations or splicing issues. ASOs offer a temporary or adjustable way to modify gene expression, making them a good choice when safety, flexibility, or the ability to fine-tune dosing is important.
Can gene therapy doses be repeated?
Yes, gene therapy treatments can occasionally be administered more than once, depending on the therapy type and the clinical guidelines in place. Certain gene therapies are specifically created to allow for multiple or follow-up doses if required. It's important to discuss with your healthcare provider whether repeat dosing is appropriate for your specific treatment plan.
What are the main long-term risks for each?
Gene therapy comes with certain risks. One major concern is insertional mutagenesis, where the therapy may unintentionally alter genes, potentially leading to cancer. Additionally, the immune system might react to the viral delivery vectors, causing inflammation. Since gene therapy often results in permanent changes, any unexpected complications could have lasting effects.
ASO therapy, on the other hand, has its own set of challenges. These include off-target effects, which may interfere with normal gene expression, and the possibility of long-term toxicity when treatments are repeated. Immune responses can also occur. Unlike gene therapy, the effects of ASO therapy are reversible if treatment is stopped. However, continued use over time might increase the risk of cumulative toxicity.



