How Compassionate Use Supports Rare Disease Patients
Explains how FDA expanded access gives investigational drugs to rare disease patients, outcomes, barriers and policy solutions.

Compassionate use programs, also called expanded access, provide a critical option for patients with rare diseases who have no approved treatments or cannot participate in clinical trials. These programs allow access to investigational drugs awaiting FDA approval, offering potential relief for life-threatening conditions. Here’s what you need to know:
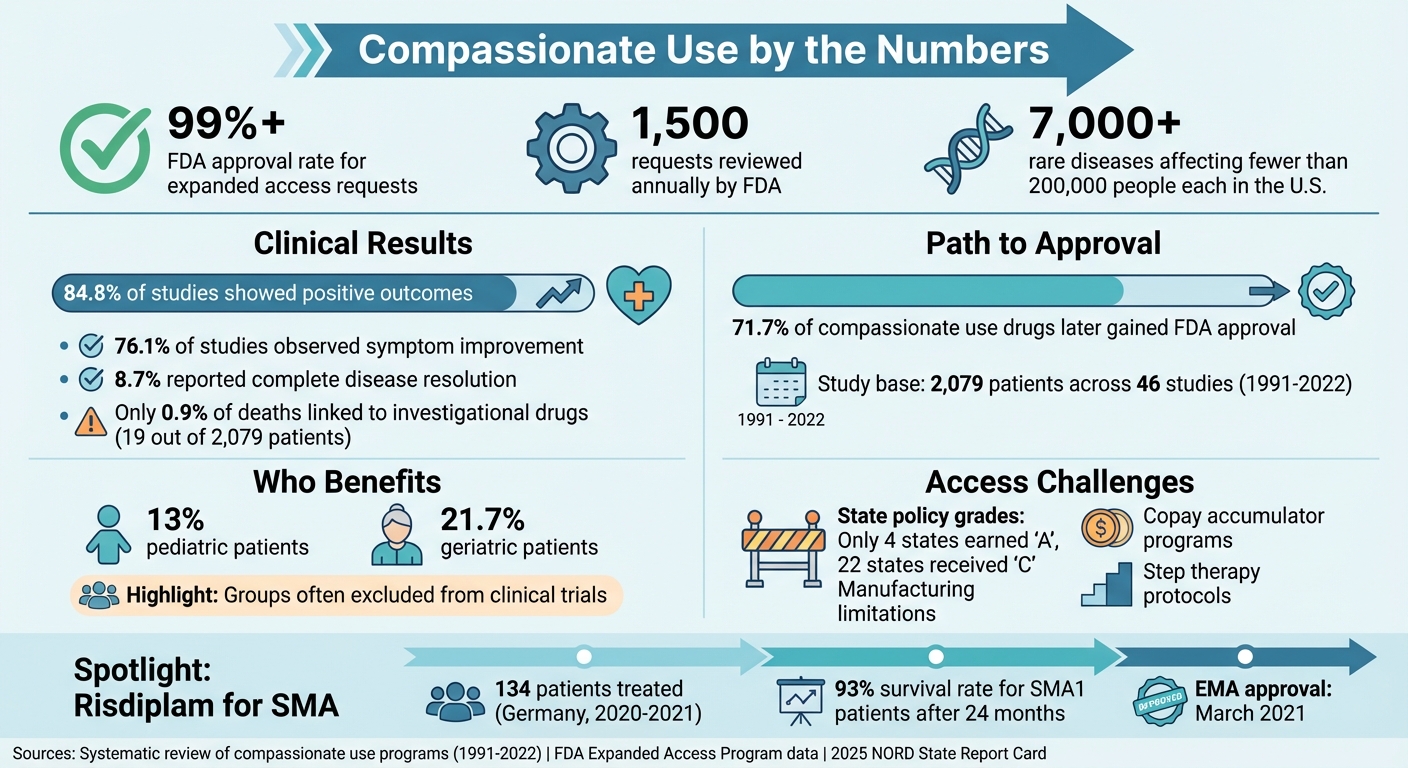
- FDA Approval: Over 99% of compassionate use requests are approved, often within days.
- Rare Diseases: With over 7,000 rare diseases affecting fewer than 200,000 people each in the U.S., many patients face limited options and strict trial criteria.
- Positive Outcomes: A review of 2,079 patients showed 84.8% experienced clinical improvement, and 71.7% of the drugs later gained FDA approval.
- Challenges: Barriers include limited drug supply, manufacturer concerns about regulatory risks, and financial hurdles like copay programs and Medicaid cuts.
While compassionate use programs offer hope, systemic changes are needed to make them more accessible and effective for rare disease patients.
Compassionate Use Programs: Key Statistics and Outcomes for Rare Disease Patients
Navigating FDA Expanded Access and Compassionate Use - Rare Disease Drug Development Series Preview
What Is Compassionate Use?
Compassionate use, also called expanded access, is a way for patients with serious or life-threatening conditions to access experimental treatments. Unlike clinical trials, which are designed to collect data under strict guidelines for regulatory approval, compassionate use focuses solely on providing treatment to patients who have no other viable options. This approach is especially important for individuals with rare diseases who might not have any approved therapies available. The FDA's Expanded Access Program formalizes this process, offering a structured framework for such cases.
FDA's Expanded Access Program
The FDA's Expanded Access Program outlines specific criteria for eligibility. To qualify, a patient must have a severe or life-threatening condition with no comparable treatment options. Additionally, the potential benefits of the experimental therapy must outweigh the risks, and granting access should not interfere with ongoing clinical trials. Each year, the FDA reviews around 1,500 requests for expanded access. However, the ultimate decision to provide the treatment lies with the pharmaceutical company.
How Compassionate Use Works
The process begins when a licensed physician determines that a patient meets the criteria for compassionate use and is willing to explore investigational treatment options. The physician must then obtain the cooperation of the drug's sponsor, as companies are not obligated to provide access to their experimental products. If the sponsor agrees, either the physician or the sponsor submits a clinical protocol to the FDA for approval.
Pharmaceutical companies are required to publicly disclose their policies for expanded access, ensuring greater transparency - an important aspect for patients dealing with rare diseases. Companies also have flexibility in determining costs. They may choose to provide the drug for free or charge only for direct manufacturing expenses, but patients are typically responsible for any related medical services.
How Compassionate Use Affects Rare Disease Patients
Survival Rates and Quality of Life Improvements
Compassionate use programs have shown measurable benefits for patients with rare diseases. A systematic review covering cases from 1991 to 2022 revealed that 84.8% of studies reported positive outcomes, with many patients experiencing either complete resolution of their disease or noticeable symptom relief. Among 46 compassionate use studies involving 2,079 rare disease patients, 76.1% of studies observed symptom improvement, while 8.7% reported complete disease resolution.
Safety data from these initiatives is also reassuring. Only 0.9% of deaths (19 out of 2,079 patients) were linked to the investigational drugs used in these programs. This low rate highlights that compassionate use can offer life-saving treatments without exposing patients to excessive risks.
One notable example comes from Germany, where a compassionate use program provided the oral drug risdiplam to 134 patients with Spinal Muscular Atrophy (SMA) types 1 and 2 between March 2020 and March 2021. These patients faced challenges with intrathecal injections due to severe scoliosis or respiratory complications. Real-world data from this program showed no new safety concerns, and the drug was granted EMA approval in March 2021. Clinical trials for risdiplam also showed promising results, with 93% of SMA1 patients surviving after 24 months of treatment.
Beyond improving survival rates, compassionate use programs also open doors to more personalized treatment options.
Personalized Drug Discovery Approaches
Compassionate use programs pave the way for customized therapies that address specific genetic mutations. In one 2025 case series, three male patients (average age 16.3 years) with Shwachman-Diamond syndrome were treated with ataluren, a drug targeting their SBDS nonsense mutations. After 12 months, two patients experienced restored protein synthesis, improved bone marrow function, normalized neutrophil counts, and a 4.6-fold increase in fecal elastase-1 levels - a significant marker of pancreatic function.
RareLabs is a leader in developing personalized therapies for rare genetic disorders. Their process involves three key steps: patient-derived cell models, multi-modality drug discovery, and regular progress updates. Using approaches like small molecule screening, antisense oligonucleotide therapy, gene replacement therapy, and combination strategies, RareLabs tailors treatments to each patient’s unique needs. This mirrors the goals of compassionate use programs, which often provide access to investigational drugs for patients who don’t qualify for standard clinical trials due to factors like age, concurrent medications, or disease severity.
The "named-patient" pathway within compassionate use ensures that patients receive the investigational drug itself rather than a placebo. This is especially vital for individuals with rapidly progressing conditions. Pediatric and geriatric patients, who are frequently excluded from clinical trials, particularly benefit from this approach. A review found that 13% of compassionate use cases involved pediatric patients, while 21.7% involved geriatric patients. These outcomes underscore the importance of tailored therapies for patients with rare diseases.
Barriers to Compassionate Use Programs
Breaking down access barriers is a must if compassionate use programs are to make a real difference for patients with rare diseases.
State-Level Differences in Access
While the FDA approves nearly all expanded-access requests, state-level policies often determine whether patients can actually benefit. The 2025 NORD State Report Card graded the U.S. a "B" overall for rare disease policy. However, only four states earned an "A", while 22 states received a "C" - highlighting uneven access across the country.
One major hurdle comes from copay accumulator programs, which block manufacturer assistance from counting toward a patient's deductible. This leaves patients with hefty out-of-pocket expenses. Eric, a California patient, shared his experience:
When copay accumulator programs are used, that assistance suddenly doesn't count toward my deductible or out-of-pocket costs. For patients like me, that can mean thousands of dollars in unexpected bills.
In 2025, states like Indiana, Iowa, Maryland, and Nevada passed laws banning these programs to safeguard patient access.
Step therapy protocols present another challenge. These policies force patients to try lower-cost medications first, which can be problematic for those with rare diseases. Some states have made progress here - New Jersey, for example, jumped from an "F" to an "A" in 2025 by reforming its policies to protect stable rare disease patients from being forced to switch treatments. Alaska also improved, moving from an "F" to a "C".
Medicaid coverage cuts have added to the strain. In 2025, Georgia reduced Medicaid eligibility for parents from 100% to just 29% of the Federal Poverty Level, leaving many rare disease families without coverage. Meanwhile, Massachusetts and Rhode Island limited retroactive Medicaid coverage, creating delays and financial stress for families. On top of that, some insurers require patients to consume at least 25 mL per hour of medical nutrition to qualify for coverage - a threshold that many fragile patients simply cannot meet.
These state-level challenges are compounded by additional barriers created by drug manufacturers.
Addressing Financial and Regulatory Obstacles
Beyond state regulations, drug manufacturers themselves often limit access. According to Theron Odlaug, Rare Disease Advocate and Operating Partner at Signet Healthcare Partners:
The FDA is rarely the barrier. The agency authorizes more than 99% of expanded-access requests it receives, typically within days. Instead, decisions rest with drug companies.
Manufacturers often decline requests due to production limitations, particularly for complex biologics and gene therapies. There’s also concern that adverse events during compassionate use might jeopardize regulatory approval. For example, in February 2026, Anna, a patient with Dravet syndrome, was denied access to EMPEROR gene therapy because of the risks involved in discontinuing her current medication.
Several strategies could help reduce these barriers. For instance, implementing data firewalls could ensure that real-world data from compassionate use programs isn’t included in pivotal efficacy analyses, easing concerns about regulatory complications. Liability safe harbors could protect manufacturers from legal repercussions tied to adverse events during FDA-authorized expanded access. Additionally, tax credits or targeted grants could help offset the high costs of manufacturing and administering investigational drugs.
Philanthropic efforts also play a crucial role. Groups like NORD provide temporary financial assistance to fill gaps when copay cards run out or insurance delays coverage, preventing treatment interruptions for conditions like metabolic disorders. As Odlaug explained:
Incentivizing compassionate use is not about undermining science. It is about recognizing that ethical leadership in rare-disease drug development includes responsibility to those left behind by trial design.
Conclusion: Moving Forward for Rare Disease Patients
Compassionate use programs serve as a critical link between clinical trials and full regulatory approval. With 84.8% of studies showing positive outcomes and over 99% of FDA expanded access requests being approved, these programs provide a lifeline for patients when standard treatments are no longer an option.
However, significant hurdles persist. Manufacturing limitations and regulatory concerns continue to restrict access, leaving many patients and their families in a state of uncertainty.
Addressing these challenges calls for teamwork and systemic changes. Solutions like implementing data firewalls, establishing liability protections, and offering tax incentives can maintain the integrity of clinical trials while enabling companies to provide expanded access without financial strain. These steps are essential for ensuring sustained progress.
As Theron Odlaug, Operating Partner at Signet Healthcare Partners, put it:
If we truly believe that patients come first, then we must build a system where hope is not confined by inclusion criteria and where companies are supported, not punished, for choosing compassion.
Organizations such as RareLabs are already paving the way with personalized drug discovery programs designed to match treatments to individual genetic profiles, demonstrating what is possible when collaboration and innovation come together.
The message is clear: breaking down barriers and creating incentives isn’t just a goal - it’s a necessity to make compassionate use a viable option for every rare disease patient.
FAQs
Am I eligible for compassionate use?
Eligibility for compassionate use hinges on specific factors, including your medical condition and whether there are approved treatments or clinical trials available. This option is often considered for patients dealing with rare diseases who have exhausted all other viable treatment options.
To pursue compassionate use, your healthcare provider must initiate a request with the drug manufacturer and regulatory agencies. Approval typically depends on meeting certain criteria, such as the severity of the disease and the absence of alternative treatments.
How do I ask a drug company for expanded access?
To begin the process of requesting expanded access, reach out to the drug manufacturer directly. Explain your medical condition and why you believe the investigational drug could benefit you. Collaborate with your healthcare provider to gather the necessary paperwork, including documentation of your medical need and a letter of support from your physician. Submit these materials to the manufacturer and, if required, the FDA. Additionally, patient advocacy groups and FDA resources can provide helpful guidance throughout the process.
Who pays for the drug and related care?
The cost of medications and associated care under compassionate use programs is typically not covered by insurance or healthcare systems. This means patients or their families often bear these financial burdens.



